Biotech Inventions and Patentable Subject Matter
3
Biotech Inventions and Patentable Subject Matter
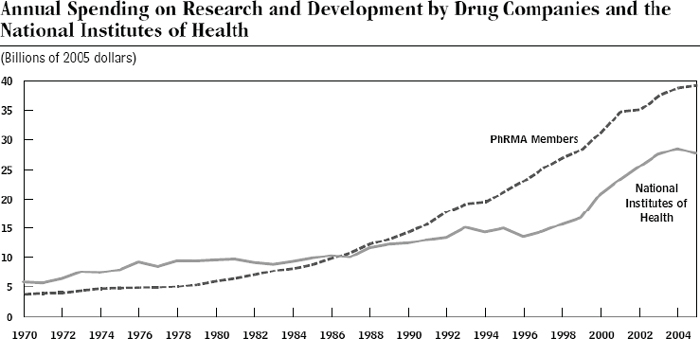
INFORMATION IS CRUCIAL in a knowledge economy based mainly on computer science, electronics, nuclear energy and biomedical technology. Biotechnology has brought about new types of drugs and agricultural technology. This chapter retraces the evolution of biotech industry and patent protection in developed countries. In comparing the US and European approaches to patentability of biotechnological inventions, the chapter attempts to explain possible ethical, economic and political concerns arising from their patent protection. The granting of overly broad patents that could potentially block further research has been one such concern. Raw sequence data, for example, should therefore be treated as ‘pre-competitive information kept in the public domain’.1 Licensing policies are another aspect for consideration. Later, in developing countries, there emerged a widespread fear that biotechnology patents would be extended to animals and plants found in nature, or traditional knowledge taken as such.2 Obviously, patents are given not to living organisms found in nature or traditionally applied knowledge as such, but to man-made inventions which fulfil the criteria of novelty, inventive step and industrial applicability.
For these complex reasons, patenting in the area of genomics requires patent owners, the law makers and enforcers to have particular responsibilities in ‘balancing the system in a way so as to strictly commensurate the scope of patent protection to the actual contribution to the art by the inventors and to deny patents whenever purely speculative “inventions” are at stake’.3 Some scholars later argued that where there is no biotech research or industry, patent protection of genes is not useful.4 However, there will be few societies which do not need any form of biotech research. For promoting international scientific or research cooperation or for encouraging new businesses in developing countries, such protection may be useful, as ownership in trade in technology encourages responsibility and cooperation.
I BIOMEDICAL RESEARCH AND GENE PATENTING
A A New Generation of Biotechnology
Biotechnology medicines utilising enzymes and protein which are produced by the human body have existed for centuries. However, in 1953 publication in the journal Nature of the discovery by JD Watson and F Crick of the DNA5 double helix structure represented the beginning of a new era of genetic engineering based on life science. In the 1970s, S Cohen6 and H Boyer7 invented a recombinant DNA (rDNA) technology. This technology allowed the coding of heterogeneous DNA, to be introduced into host organisms (such as microorganisms or cell lines) by appropriate vectors (such as plasmids and viruses). If genes to be transformed come from different biological species, the resulting genetically modified organisms (GMOs) are transgenic, while GMOs without DNA from other species are cisgenic. Modified or novel DNA is expressed in transformed host organisms and translated into a protein. For biotechnology to be useful for health purposes, gene expression and protein formation in the human body, and their extraction, purification and manufacturing are necessary. This, in turn, requires further scientific knowledge and biotechnology such as cell culture, genetic modification and gene cloning.
In 1985, polymerase chain reaction (PCR) technology was invented by K Mullis and was patented by his employer company Cetus, later Chiron, and sold to Hoffman-la-Roche.10 PCR enzymatically amplifies short segments of DNA and nucleotides by using single-stranded DNA as a template, and DNA oligo-nucleotides (also called DNA primers) which are required to initiate DNA synthesis. PCR and its improved technologies (such as those using Taq thermostable DNA polymerase) were created to perform a wide range of genetic manipulations and contributed not only to the advancement of genomics and molecular biology, but also to the establishment of new methods of drug, diagnostic and therapeutic discoveries.
In 1989, President Bush proposed the idea of discovering each of the estimated 20–25,000 human genes and making them accessible for biological study. The following year the Human Genome Project, a US-led international team of 350 scientific institutions, was established. The Department of Energy’s Human Genome Program and the National Institute of Health (NIH) and National Human Genome Research Institute (NHGRI) jointly sponsored the Human Genome Project. Known as the HGP, this became an international effort which continued for 13 years from the time it formally began in October 1990 until it was completed in 2003. Another goal of this project was to determine the complete sequence of the 3 billion DNA subunits (based on the human genome). As part of the HGP, parallel studies were carried out on selected model organisms, such as E coli bacteria11 and mice, to help develop technology and interpret human gene functions.
Ten years later, on 26 June 2000, the international consortium declared that the HGP was almost complete. R&D investments in genomics and gene technology have led to the discovery of new genes, screening of drug targets and lead-compounds, and subsequent clinical studies. Patent protection created a scheme to recover these costs and help the US lead genome-based drug discoveries.
Increased knowledge of genes, together with recombinant DNA (rDNA) technology, came to be used in a wide spectrum of agricultural, biological and medical research and production, and for the discovery of pharmaceutical drug targets and exploring gene therapy. Drug development which once relied mainly on chemistry has since shifted to controlling biological systems using biological material (such as genes, protein, antibodies, cells, and genetically modified animals and plants) and biotechnology.
B Spread of New Technologies
The advent of genomics, genetics, and proteomics has created massive opportunities but also poses challenges to university researchers, pharmaceutical companies and regulators alike. For drug discovery and development, the paradigm change in the late 1990s was radical. A wide range of new in-vitro technologies and techniques for animals and humans replaced traditional chemical manipulation, requiring not only more sophisticated, massive investments, but also further education in science, basic research, and biotechnology. For companies, increased regulatory requirements in both the pre- and post-launch periods resulted in significant changes in risks and benefits. For regulators, the need to ensure non-toxic, safe and effective drugs has led to significant delays in developing new criteria for judging whether medical inventions submitted for examination are indeed safe and effective.12
Medicinal compounds can be therapeutically effective when they inhibit or change the behaviour of pathogens (such as bacteria and viruses which cause diseases) or by blocking the metabolism of pathogens. They can also be effective by selectively targeting disease-causing organisms such as receptors or cells. When conducting biomedical research, receptors, enzymes and proteins became important targets of screening to discover possible drug candidates. Pharmacogenomics, a new field of drug designing, discovery and development, was made possible by using information on the human genome. This, in turn, gave rise to a new therapeutic method which dealt with the genetic causes of diseases and tailor-made therapy by taking into account individual variations.
Recombinant DNA technology also became indispensable in inventing research tools for life science such as agricultural, biological research and drug discovery. Regardless of whether it is a commercial product or not, a research tool is any instrument, information or technology which can be used by laboratory researchers in the fields of life science or drug discovery. In addition to genetic information such as receptors, analytical methods (such as PCR methods, screening methods, rDNA technology) and devices (such as PCR devices, DNA chips, and DNA sequencers), and biotechnology-related informatics (such as gene sequence databases, protein bioinformatics, gene libraries, and combinatorial chemistry) also became necessary research tools for R&D. Laboratory animals such as transgenic animals, with insertion or deletion of genes for specific diseases, came to be frequently used.
For the spread of rDNA technology and innovation of technology, licence terms became vital. Licensing conditions differ considerably among different biomedical research tools. Cohen-Boyer rDNA technology13 and the polymerase chain reaction (PCR) technology14 are often contrasted, iconic illustrations.15 The question of whether any licence terms promote R&D and technological innovation depends on the scope of patentable subject matter, the technical fields concerned and the structure of the R&D market (such as the number of competitors). Cohen-Boyer cloning technology as well as the PCR became indispensable for the further advancement of life science, but the ways in which these technologies were licensed differed considerably. Cohen-Boyer cloning patents were granted to Stanford University and the University of California, both of which granted non-exclusive licences for low royalties to Genentech (founded by Boyer) and hundreds of other companies.16 These universities used this type of licence revenue as a source of income for their basic research, which contributed to the spread of technology and to R&D. Kohler and Milstein were the first to obtain patents for murine monoclonal antibodies.17 However, they did not claim for rights to each individual antibody with respect to each individual antigen which resulted from their research. Just like the Cohen-Boyer patents, these research tools were licensed at reasonable prices, which led to significant achievements. The Keller and Milstein licence terms contributed also to the successful commercialisation and widespread use by researchers of their technologies, and are said to be the most successful examples of university technology transfer, generating revenue and leading to a range of new products.
The use of PCR technology, by contrast, was conditioned on the payment of reach-through royalties, even on PCR products and the purchase of Roche reagents. A reach-through agreement is an agreement which requires the payment of royalties as a fixed percentage of sales of the licencee products, which are developed using the licenced technology. Research tools without substitute technology, combined with network effects, became a controversial R&D issue.
In the process of analysing the human genome, researchers and venture companies have continued to apply for patents for genes using the Patent Cooperation Treaty (PCT) route (see chapter 2). Not only are DNAs chemical substances, they have specific characteristics of carrying information. For example, receptors conveying information regarding intruding entities (which could play the role of a doorkeeper for the cell entry of viruses, etc). As there was often no prior art for this type of chemical substance, patents could be granted relatively easily. In the early stages of drug discovery, it is possible to determine whether specific genes would become screening materials as drug targets. When gene patents with broad claims were granted, there were possible problems in relation to whether this would have any effect on pharmaceutical research and development and future industrial structures. Gradually, debate has grown on how substitutable this type of information is and whether the protection of such information by patents will have any effect on pharmaceutical R&D.
II PATENT PROTECTION OF BIOTECH INVENTIONS
A Expansion of Patentable Subject Matter
With the advent of biotechnology, the discussion arose as to whether and how products of nature could be patented. In the 1980s a groundbreaking decision that expanded patentable subject matter was handed down. In Diamond v Chakrabarty,18 the US Supreme Court decided that genetically engineered plasmids,19capable of decomposing crude oil into multiple components, are patentable, and found that such microorganisms could be patentable.20 Section 101 of the US Patent Act defines patentable inventions and stipulates that: ‘whoever invents or discovers any new and useful (a) process, (b) machine, (c) manufacture, (d) composition of matter, or (e) any new and useful improvement thereof, may obtain a patent, subject to the conditions and requirements in the law’.21 Items (b) to (d) are products, as opposed to (a) which is a process. Sections 102 and 103 set out those conditions for patentability, which are novelty, and for loss of right to patent, and non-obvious subject matter. The inclusion of the word ‘useful’ in section 101 shows that there is no pre-emption from a public perspective. The Supreme Court has found limits to section 101, expressed as ‘abstract ideas’, ‘laws of nature’22 and ‘natural phenomena’. The Court later stated that not all discoveries are patentable, independently of the question of whether they are products or processes.23
In Chakrabarty, the Supreme Court interpreted section 101 to mean that the decision of whether to grant patents depended not on whether the subject matter was a living thing (a product found in nature) or not, but whether or not the subject matter of the claims were human-made inventions, in the form of process, machine, manufacture, or composition of matter. According to the Court, ‘anything under the sun that is made by man’ that is not an abstract idea, law of nature, or natural phenomenon which fulfils the conditions delineated in sections 102 and 103, as well as other requirements under the US Patent Act, is patentable. The patent claims in Chakrabarty covered: (i) a method of producing bacteria; (ii) an inoculum comprised of a carrier material floating on water; and, (iii) the genetically modified bacteria itself. According to the Supreme Court, the microorganism in Chakrabarty was ‘a product of human ingenuity “having a distinctive name, character [and] use” and research’.24
The petitioner in Chakrabarty argued that the legislature should determine the patentability of genetic technology, which was unknown when Congress enacted section 101. The Court, however, held that these provisions were written in broad terms to fulfil the constitutional and statutory intention of promoting ‘the progress of science and the useful arts’, which encompassed all possible meanings for social and economic benefits, and a legislative or judicial fiat as to patentability would not hinder scientific probing into the unknown. This judgment recognised that patents could be granted not only for microorganisms but also for certain living things under specific conditions, and worked to promote biotechnology and subsequent breakthroughs in the US.
the rule that the discovery of a law of nature cannot be patented rests not on the notion that natural phenomena are not processes, but rather on the more fundamental understanding that they are not the kind of ‘discoveries’ that the statute was enacted to protect. The obligation to determine what type of discovery is sought to be patented must precede the determination of whether that discovery is, in fact, new or obvious.26
The Chakrabarty judgment by the US Supreme Court gave a decisive signal for venture capitalists to pour money into the scientific efforts of predominantly academic researchers and led to the rise of a new branch of industry – the biotechnology industry.27
B Genetically-engineered Animals and Plants
Since the 1970s, biotechnology has been one of the factors leading to innovations in the agricultural and pharmaceutical R&D. Inventions relating to recombinant DNA technologies on plants, animals and microorganisms began to be protected in different countries under patent or plant varieties protection laws. In the late 1980s, it was still only in the US, Switzerland, Japan and a few European countries that biotechnology was used for industrial purposes. Yet the globalising market made it necessary to harmonise the conditions in which patents are granted to promote biotechnology inventions. Patent protection efficiently expands the market beyond the domestic market to make global competition possible.
To grant a patent, an invention must be disclosed through a written description. If an invention involves a microorganism28 or the use thereof, disclosure may be made only by providing a sample. For this reason, the Budapest Treaty on the International Recognition of the Deposit of Microorganisms for the Purposes of Patent Procedure was adopted in 1977 (amended in 1980). The Contracting Parties29 are required to recognise, for the purposes of patent applications, the deposit of microorganisms with any ‘international depositary authority’. This description refers to any scientific institution capable of ‘culture collection’ of samples of microorganisms, irrespective of whether such authority30 is within or outside the territory of that state (on the TRIPS Agreement and microorganisms see chapters 5, 10 and 12).
In the US, biotechnology inventions relating to animals were recognised as patentable, and since Ex parte Hibberd31 in 1985, patentability of those inventions concerning plants was also recognised. In Japan in the 1980s, a number of administrative and court decisions confirmed that gene-engineered animals and plants (microorganisms in 1981, plants in 1985, and animals and living organisms in 1991), as well as the processes to produce them, and new varieties of plants could be protected by patents, under the Plant Variety Protection and Seed Act. In Europe, harmonising patent protection of biotechnology inventions encountered more resistance.
C EPC Statutory Exceptions and Biotechnology Inventions
i Before the European Patent Convention
In Europe, the path to establishing a system for protecting biotech inventions has been long and not linear. Since the late 1960s, Member States of the European Community started to grant patents to protect biotechnology inventions. In 1969, the German Federal Supreme Court (BGH) in its Red Dove (Rote Taube) decision,32 recognised in principle the eligibility of inventions relating to animals and breeding methods for patent protection, provided that the technique could be reproduced with sufficient prospects of success. The Court however did not confirm the patent in question, due to the lack of repeatability in this case. Eighteen years after the German Federal Supreme Court decision on Red Dove, the same Court, in the ‘Rabies Virus’ decision,33 accepted that deposits of biological material (microorganisms) were a complement to the written description and as an enabling disclosure.
In the meantime, the European Patent Convention (EPC) was adopted in 1973. Its Article 53(b) excludes from patent protection essentially biological processes for the production of animals and plants. Thus, a breeding method, such as crossing and selection, which existed in the Red Dove decision, became ineligible for patentability. On the other hand, non-essentially biological methods for the production of animals and plants remain patentable, as do inventions related to animals and plants, if their feasibility is not limited to a plant variety or animal race.34
ii EPC and Patentability
Chapter I of the EPC concerns ‘patentability’, under which Article 52 EPC defines patentable inventions. According to Article 52(1), ‘European patents shall be granted for any inventions, in all fields of technology,35 provided that they are new, involve an inventive step and are susceptible of industrial application. Article 52(2) however notes that the following shall not be regarded as inventions within the meaning of Article 52(1): (a) discoveries, scientific theories and mathematical methods; (b) aesthetic creations; (c) schemes, rules and methods for performing mental acts, playing games or doing business, and computer programs. According to Article 52(4) EPC 1973, ‘methods for treatment of the human or animal body by surgery or therapy and diagnostic methods practiced on the human or animal body shall not be regarded as inventions which are susceptible of industrial application within the meaning of paragraph 1. This provision shall not apply to products, in particular substances or compositions, for use in any of these methods.’ This provision was deleted by the EPC 2000 and its content transferred to Article 53 (c) of the EPC 2000 as one of the matters in respect of which European patents shall not be granted.
Notable for comparative purposes is that the EPO generally applies the problem-solution approach in assessing the requirement of inventive step.36 The problem-solution approach was developed during the 1980s by the Technical Board of Appeals (TBA) and generally applied today by the Examining Divisions, the Opposition Divisions, and the TBA of the EPO. This approach consists of defining the technical problem to be solved by the proposed invention, identifying the closest prior art in view of which the technical results of the proposed invention is assessed for determining whether a person skilled in the art would have suggested the claimed technical features for the solution provided by that invention.
Whereas the US patent law does not pre-empt patentability on the public or morality grounds or specifically for agricultural concerns, the EPC 1973 and 2000 (and the subsequent EU Biotech Directive) provide for exceptions to patentability in Article 53.37 According to Article 53 of the EPC, European patents shall not be granted in respect of: (a) inventions the commercial exploitation38 of which would be contrary to ordre public or morality; (b) plant or animal varieties or essentially biological processes for the production of plants or animals; this provision shall not apply to microbiological processes or the products thereof; (and (c) ‘methods for treatment of the human or animal body by surgery or therapy and diagnostic methods practiced on the human or animal body’ was added in the EPC 2000, as described above39).
These exceptions can be traced back to the circumstances of the time when the EPC was adopted in 1973. Article 53(b) of the EPC excludes ‘plant or animal varieties or essentially biological processes for the production of plants or animals’, whereas microbiological processes and the products thereof are patentable under this provision. The main reason at this time for these EPC exclusions was the existence of other international treaties. As for the protection of plant varieties, the International Convention for the Protection of New Varieties of Plants (UPOV Convention)40 was adopted in 1961 in Paris (see chapter 2). This Convention regarded the protection of new varieties of plants solely as an ‘agricultural issue’, and prohibited dual protection by providing that a contracting party may provide only one form of protection for one variety. This provision was lifted when the Convention was revised on 19 March 1991, but Article 53(b) of the EPC, which was signed in 1973, continued to exclude plant varieties from the patentable subject matter.
Later, the Technical Board of Appeal (TBA) of the European Patent Office (EPO), in Ciba-Geigy41 and Lubrizol,42 held that plants themselves taken as a genus claim, as opposed to a ‘variety’,43 were patentable. Subsequently, dozens of patents were granted on plants. However, the TBA Decision in PGS44 in 1995 held that plants and plant varieties including the genus claim were not patentable, in accordance with the provisions of Article 53(b) of the EPC 1973. These decisions took into account the European Community Plant Varieties Regulation,45 which came into force in 1994.
Following this, in 1999, the Enlarged Board of Appeal of the EPO decided in a case relating to Transgenic Plant/Novartis II46 that plants which are products of rDNA technology avoid the prohibition on patent grant under Article 53(b), if specific plant varieties are not individually claimed. This decision indicated that, even if plant varieties themselves are not patentable, patent granted to a certain invention might cover a plant variety as well. The Board was probably conscious of the draft Biotechnology Directive of the European Community (EC), which proposed that a concept of genetic engineering applicable to more than one variety was an invention.47
Patentability of animal varieties was discussed during the negotiations for the Strasbourg Convention on the Unification of Certain Points of Substantive Law on Patents for Invention (Law on International Patent Treaties) which was adopted in 1963, two years after the UPOV Convention48. Participating countries debated fiercely the ethical implications of patenting animal varieties, and no agreement was reached. Article II of the Strasbourg Convention, therefore, allows Contracting Parties not to protect plant or animal varieties or essentially biological processes for the production of plants or animals, except for microbiological processes or the products thereof. The EPC exclusion of plant and animal varieties derives from this provision of the Strasbourg Convention.
In general, the exclusions from patentability in Article 53 of the EPC 1973 have been construed narrowly. In Europe, patent protection of biotechnological inventions relating to animals was resisted from various perspectives, such as ethical, religious, traditional values, animal protection and farmers’ rights perspectives.49 In the Harvard OncoMouse case of 1990, the Examination Division of the EPO initially rejected the patent application in 1989, on the grounds of above-mentioned Article 53(b) EPC. The OncoMouse is a laboratory mouse, genetically modified and designed to carry a specific gene which increases the mouse’s susceptibility to cancer.50 The applicant appealed against this decision (T 19/90) and the Board of Appeal of the EPO held that the exception to patentability under Article 53(b) EPC applied to certain categories of animals, but not to animals as such; and that, in the absence of serious doubts substantiated by evidence, there was no reason to reject the application on the ground that it involved an extrapolation from mice to mammals in general.51 According to the Board, the exception to patentability under Article 53(b) EPC should be construed narrowly, and a test should have to be performed on a case-by-case basis. In terms of ordre public or morality under Article 53(a) EPC, the Board stated that the suffering of animals and possible risks to the environment on the one hand, and the invention’s usefulness to mankind, on the other, must be weighed up. The decision to grant a patent was dated 3 April 1992 and took effect on 13 May 1992, the day it was mentioned in the European Patent Bulletin. In this case, ‘animal varieties’, ‘races animales’ and ‘Tierarten’, whose scientific meanings differ, were used in the three authentic languages of the EPC and remained undefined.
A patent was finally granted,52 but 17 oppositions were filed against it based, in particular, on Article 53(a) and (b) EPC. On 7 November 2001, the Board of Appeal limited the claims to rodents (T 315/03)53 under which animal variety is only a subunit.54 While science is evolving and uncertain, and technology is rapidly moving, patent protection of biotechnological inventions has remained controversial in Europe. In the years leading to the adoption of European Community Directive 98/44/EC of 6 July 1998 on the legal protection of biotechnological inventions (EU Biotech Directive)55, particularly, questions relating to patent protection of biotech inventions were intensely discussed.56
D EU Biotech Directive
Each European country had its own approach to patentability, reflecting in part the agricultural or industrial focus of their individual economies and particular values. These countries also employed different legal formulations, concepts and interpretations in order to conceptualise biotechnology inventions and when to grant patent protection. Where biotechnology based on life science was concerned, different religious and ethical values also made the issue more complex. Among Member States of the EU (European Community (EC) at that time), legislation, implementation and interpretation of domestic laws concerning the patentability of genetic manipulation or parts of the human body varied and lacked legal predictability and certainty. This was the aim of the draft Directive proposed in 1988 by the European Commission. This was proposed with a view to harmonising57 national laws, promoting commercialisation of biotech R&D results, and clarifying the scope of patent protection of biotechnology inventions, across EC Member States. Biotechnology inventions were becoming socially useful, but raised issues not only for legal harmonisation, but also relating to ethical norms which some felt could be infringed.
Inventions of a technical character which are new, having inventive step and industrial applicability, are patentable in the EC Member States, and genes which fulfil these requirements and which are found in nature may be patentable, provided that they are isolated from the human body or produced by means of a technical process.58 The US Supreme Court in Chakrabarty noted that the patentee, by his inventive activity, produced a new and useful bacterium with markedly different characteristics from any found in nature.59 Under the EU Biotech Directive, genes found in nature are patentable, if isolated, purified and classified, and reproduced outside the human body, even if the product found in nature has identical functions.60 A mere genomic DNA sequence is not considered to be an invention, unless it is isolated from nature and sequenced. The inventor must also indicate the method for its repeatable production, in fulfilment of the general rule for patentability.
In the EU Biotech Directive, respect for dignity and integrity of the person is the basic ethical rule wherever such a rule is relevant to biotechnology inventions, as Recital 16 of the Directive states:
. . . patent law must be applied so as to respect the fundamental principles safeguarding the dignity and integrity of the person; whereas it is important to assert the principle that the human body, at any stage in its formation or development, including germ cells, and the simple discovery of one of its elements or one of its products, including the sequence or partial sequence of a human gene, cannot be patented; whereas these principles are in line with the criteria of patentability proper to patent law, whereby a mere discovery cannot be patented.61
For ethical considerations, Article 6 of the EU Directive notes possible exceptions to patentability of those inventions whose commercial exploitation would be contrary to ordre public or morality, in particular: (a) processes for cloning human beings; (b) processes for modifying the germ line genetic identity of human beings; (c) uses of human embryos62 for industrial or commercial purposes; and (d) processes for modifying the genetic identity of animals which are likely to cause them suffering without any substantial medical benefit to man or animal, and also animals resulting from such processes. Article 6 separates the granting of patents for the results of research and development, and the commercial exploitation of that technology. This is the reason why this provision states that exploitation shall not be deemed to be contrary to ordre public or morality ‘merely because it is prohibited by law or regulation’.
The ways in which the EU Biotech Directive is construed differ significantly from the US Code. The Directive not only provides statutory exceptions such as Article 4, but also states explicitly that only ‘inventions’ are patentable. In the US, exceptions to patentability are decided judicially.
According to Article 1 of the Biotech Directive, EU Member States shall protect under national patent law biotechnological ‘inventions’. Article 3(1) stipulates the principle that ‘inventions which are new, which involve an inventive step and which are susceptible to industrial application shall be patentable, ‘even if they concern a product consisting of or containing biological material63 or a process by means of which biological material is produced, processed or used’. Article 3(2), which clarified the specific conditions in which natural products could be patented, provides that ‘biological material which is isolated from its natural environment, or produced by means of a technical process, may be the subject of an invention even if it previously occurred in nature’. This condition is also required for biological material which previously occurred in nature.64 Thus, the EU Biotech Directive confirms the patentability of naturally occurring biological material defined in Article 2(1).65
Articles 4 to 6 provide statutory exceptions to patentability. Article 4(1), like Article 53(b) of the EPC, excludes from patentability: (a) plant and animal varieties, and (b) essentially biological processes for the production of plants or animals. However, Article 4(2) of the Directive explicitly provides what is not found in the EPC; namely: ‘inventions which concern plants or animals shall be patentable if the technical feasibility of the invention is not confined to a particular plant or animal variety’. Article 4(2), confirming the Transgenic/Novartis II decision,66 makes patent protection possible for a plant or animal variety, unless specific varieties are individually claimed. This was already the case in many EC Member States, which Article 4(2) made explicit. However, it created a certain discrepancy with regard to the text of Article 53(b) of the EPC, even though the case law suggests such an interpretation. On 16 June 1999, the EPO Administrative Council adopted the Regulation,67 so that the content of the EU Biotech Directive would be reflected on the EPC. Article 4(3) mentions the patentability of inventions which concern a microbiological or other technical process or a product obtained by means of such a process.
The EU Biotech Directive in Article 5(1) distinguishes a ‘simple discovery’ from a ‘patentable invention’ with respect to elements of the human body,68 including the sequence or partial sequence of a gene. According to Article 5(2), an element isolated from the human body or otherwise produced by means of a technical process, including the sequence or partial sequence of a gene, may constitute a ‘patentable invention’, even if the structure of that element is ‘identical to that of a natural element’. Article 5(3) stipulates disclosure obligations, at the time of patent application, in relation to the industrial application of a sequence or a partial sequence of a gene.69
Articles 8 and 9 of the Directive concern the extent to which patent protection is given to biological materials possessing specific characteristics. Such protection is extended to any biological material derived through propagation or multiplication in an identical or divergent form and possessing those same characteristics. However, Article 11 derogates from this extended protection in cases where plant propagating material has been sold (or commercialised in other ways) to a farmer by the holder of the patent with his consent for agricultural use. In this case, the farmer is authorised to use the product of his harvest for propagation or multiplication by his own farm, to the extent provided for under Article 14 of Regulation (EC) 2100/94 on Community plant variety rights.70
E Examination Guidelines for DNA Sequences
Although the differences between the US and Europe resided mostly in the formulation of laws, there were also delicate differences in the criteria by which to consider to what extent biotech inventions are patentable, where human creativity is found, how repeatability of the invention (utility) is shown, in what cases separated DNA and protein structures can be said to constitute an inventive step (non-obviousness), and whether a patent right covers proliferation of gene-engineered animals and plants.
Even today, Europe, the US, and Japan seem to have slightly different views in determining novelty, inventive step, and industrial applicability (utility). Most differences reside in the timing of patent examination and thoroughness with which examiners investigate prior art inventions, according to the JPO study on the differences of inventive step.71 However, there remain also certain minimum substantive differences in the criteria of measuring inventive step, such as the example below.
In the US, in the early period of DNA-related inventions, the CAFC recognised non-obviousness (inventive step) with respect to a DNA invention specified by a DNA sequence (Re Deuel72). The Deuel case relates to an invention of DNA molecules encoding ‘human and bovine heparin-binding growth factors’ (HBGFs). A USPTO examiner rejected the non-obviousness of some claims in the patent application in Deuel, citing prior art literature.73 The USPTO Board of Patent Appeals and Interferences also determined that the cloning method74 (to facilitate the handling through the amplification of the subject gene sequences in experiments to produce gene clones) used in this invention was ordinary, and that if a specified protein sequence is public knowledge, so are its genes.
In the appeal against this judgment, the CAFC held that even if an applied cloning method is obvious, DNA with a specific sequence of encoding proteins which have actually been obtained is not obvious (ie, there is an inventive step). In this case, the CAFC based its judgment on the idea that even if an amino acid sequence for a protein is known, the number of types of DNA sequences corresponding to that protein is high. Therefore, prior art implying an amino acid sequence for a certain protein will not necessarily make DNA molecules encoding that protein obvious. The genetic code between protein and DNA does not overcome the deficiencies of cited references, and a partial HBGF amino acid sequence already disclosed by the EPO is not a sufficient description. Unless there is prior art which suggests that such amino acid can lead to a particular DNA and be prepared, such a DNA cannot be considered obvious.
This ruling of the CAFC led to many criticisms that the US had established lax standards for non-obviousness with respect to biotechnology inventions. In Japan and EPO jurisdictions, where cloning methods are known, it is considered that if the amino acid sequence of an encoded protein is known, no inventive step is present, because a person skilled in the art could easily clone the subject gene. Thus, there have been some noticeable differences in applying the criteria of patentability between the US on the one hand, and the EPO and JPO, on the other, although the differences have been minimised by cooperation among these agencies. In Re Kubin,75 however, the CAFC re-examined its own approach to ‘obvious-to-try’ tests in view of the of the Supreme Court criticism in KSR76 and its own understanding of these tests in Re O’Farrell.77 It concluded that the claimed invention (isolation and sequencing of the gene for natural killer cell activation-inducing ligand (NAIL) and the corresponding protein) was obvious. According to the CAFC, the prior art in this case teaches a protein of interest, a motivation to isolate the gene coding for that protein, and illustrative instructions to use a monoclonal antibody specific to the protein for cloning this gene, which is a well-known and reliable cloning and sequencing technique. The claimed invention, therefore, was judged as a product ‘not of innovation but of ordinary skill and common sense’. The Kubin judgment has brought the US biotechnology patentability criteria closer to those of Europe and Japan, although the US ‘obviousness’ standards still seem somewhat lower than those of the European and Japanese Patent Offices.
III COMMERCIALISATION OF BIOTECH MEDICINES
A First-Generation Biotech Medicines
Early examples of biotech medicines developed mostly in biotechnology allowing for extraction, cloning and mass-production of bioactive substances (proteins) in the body, such as human insulin – by bacillus Interferon α (which has therapeutic effects for cancer and hepatitis C, and belongs to a cytokine). Cytokines are a large and diverse family of polypeptide regulators produced throughout the body by cells of diverse embryological origin and encompassing proteins, peptides and glycoproteins. They are signalling molecules like hormones and neurotransmitters and are used for communication and interaction among cells. They regulate reactive adjustments such as immune or allergic reactions. In contrast to small-molecule chemical medicines, biotech medicines are large, complex and unstable protein molecules containing polysaccharide or oligosaccharide, and are capable of replacing or supplementing, and respond to therapeutic needs unmet by small-molecule medicines.
Erythropoietin is another example of a first generation biomedicine. It is also a cytokine, a glycoprotein hormone produced mainly by the renal cortex which controls red blood cell production and was discovered by French scientists in the early 1900s. Renal failure causes abnormal haemoglobin levels which can provoke anaemia. Later, in the 1980s, through a series of clinical trials and cloning technique inventions78 developed in the US, Amgen (α-EPO) and the Genetics Institute (GI) (β-EPO) began producing this hormone. In 1989, the US Food and Drug Administration approved the hormone, which was called Epogen. More recently, a novel protein (an erythropoiesis-stimulating protein, NESP), with anti-anaemic capabilities and a longer terminal half-life than erythropoietin, has been produced.
Biotechnology has been a fast growing sector of both science and industry. According to the European Commission Report in 2004,79 the worldwide biotechnology-based products market had grown at an annual average rate of 15% to reach approximately € 30 bn in 2000 of which biopharmaceuticals made up € 20 bn. Biotech medicines in 2000 accounted for less than 5% of the total pharmaceuticals market but were growing at 2.5 times its overall growth rate.
Table 3.1: Reported worldwide indications and sales of the top 15 biotechnology medicines (2000)
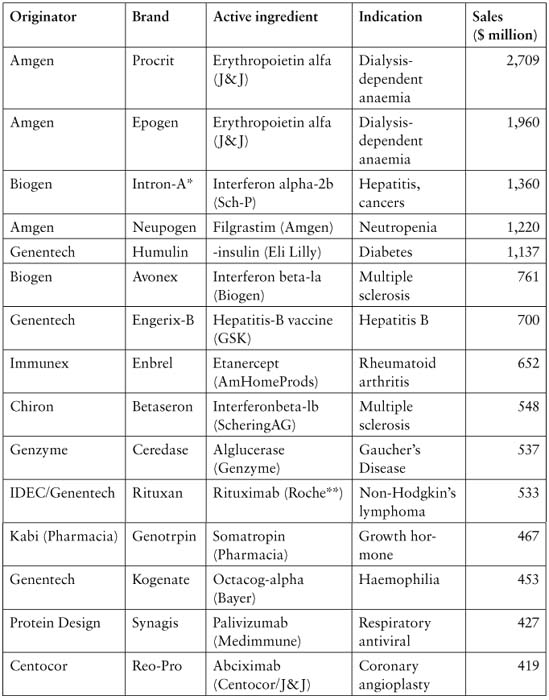
Today, the basic gene patents for the early biotech medicines have expired or will soon end, and generic versions of the original medicines (called ‘biosimilars’ in the EU; ‘follow-on biologics’ in Japan80 and the US) have started to receive market authorisation in the EU. In the EU, the European Medicines Agency (EMEA) adopted the first positive opinion for a similar biological medicinal products in early 2006, and as of February 2009, 11 products were approved.81
A few developing countries, such as India, China and Cuba, are producing follow-on biotechnology medicines.82 The Indian regulator has approved production and marketing of 20 biogeneric medicines.83 India’s exports of healthcare biotech medicine during 2007 amounted to 15.95 billion Rs and were estimated to grow to 47 billion Rs by 2012.84 Most of these products are said to be sold abroad.
As of May 2009, more than 3,000 medicines were approved as natural product-based medicines by China’s regulatory agency, the State Food and Drug Administration (SFDA).85 By what criteria these medicines are designated as natural product-based medicines is not clear.
Cuba’s product portfolio combines off-patent products (Hepatitis B vaccine, interferons, EGF) and innovative compounds (streptokinase, synthetic Hib vaccine, Heberprot P), and registered in 111 countries in total: HB vaccine 40, Interferon 34, Streptokinase 28, GAVAC 4, Hebertrans 3, EGF 2 (see chapter 11).86
B Patenting Biotech Medicines Inventions and Enforcement
In addition to patent offices, courts have adopted different policies for patentability criteria of biotechnology inventions. For example, there had been much debate in the area of biotechnology over whether it was possible to claim that something which goes beyond a natural product (for instance, a protein altered or modified to add, insert, delete or substitute amino acid residue). For example, tissue Plasminogen activator (tPA) catalyses the conversion of plasminogen to plasmin, which is the major enzyme responsible for clot breakdown. TPA can be produced by rDNA technology to be used to block this conversion process and thus to treat fibrinolysis (the physiological breakdown of blood clots) and acute myocardial infarctions (AMI or heart attacks). In Japan, the 245-Met-tPA case tested whether it is possible to expand the scope of rights beyond genetically modified tPA. The case involved a dispute between Sumitomo Pharmaceuticals, which produced Met-tPA, and Genentech, which produced tPA.87 Of the 459 amino acids in these two activators, only one amino acid was different. If the language in the claims was given a strict interpretation, Sumitomo’s Met-tPA would have been considered an improvement. The Osaka District Court gave the scope of the patent a literal interpretation and ruled that the Sumitomo patent did not infringe the Genentech patent. However, the Osaka High Court overturned this decision by applying the doctrine of equivalents, and ruled that the Sumitomo patent did infringe the Genentech patent.88
In 1987, Amgen brought an action for alleged infringement by Genetics Institute (GI) and its licensee, Chugai Pharmaceutical Company. Amgen argued its case in the US federal district court and the ITC (for an injunction against exportation from Japan to the US), and the GI side made a counterclaim in the federal court. However, in 1991 the CAFC affirmed the federal district court’s judgment on the infringement of Amgen’s patent ‘008 and ruled that GI’s patent ’95 was invalid.90 Amgen has taken a large share of the EPO market in the US. In 1992 in Japan, Kirin and Chugai opted for a settlement with Amgen.
Later, TKT developed a process for making erythropoietin involving gene activation (GA-EPO) by introducing a promoter DNA inside human cells (according to TKT, GA-EPO is manufactured by the expression of GA-EPO using DNA sequences inside human cells. Amgen asserted that GA-EPO infringed the claims of its patents (production of erythropoietin by recombinant DNA technology) in several countries. The central question was whether the TKT technology was included in the claims by Amgen, ie, GA-EPO is exogenous to the cell and therefore switching on the endogenous EPO gene does not constitute infringement.
In the US, the federal district court of Massachusetts applied the doctrine of equivalents and decided that TKT had infringed five Amgen patents. The CAFC upheld this claim construction and said the fact of being ‘uniquely characterized’ by the expression of exogenous DNA sequences did not impel the court to accept TKT’s position when the asserted claims did not contain such an express limitation. However, it remanded the federal district court on several other points.91 In its reversal of the federal district court judgment,92 the CAFC ruled that Amgen’s ’422 patent was not invalid and it applied the Cybor rule,93